Good news! However, when you Google for the title of this paper several search results suggest there have been similar reports since about 2022 of a number of such ultrapotent human antibodies.
From the significance and abstract:
"Significance
Zika virus causes microcephaly in fetuses and no vaccines or therapeutics currently exist against it. Mature and immature flavivirus particles are infectious.
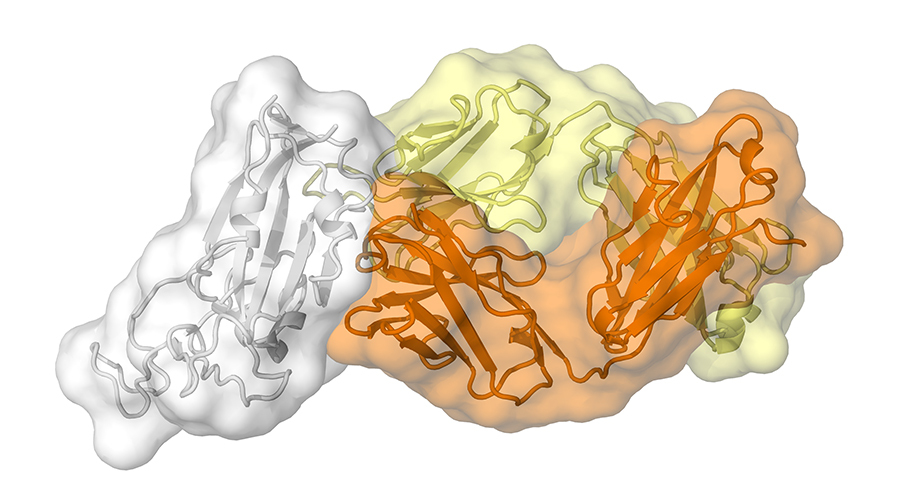
Here, we showed the cryoelectron microscopy (cryoEM) structures of an ultrapotent A9E human antibody, complexed with both mature (mZIKV) and immature (immZIKV) Zika virus, and the antibody neutralization mechanism. One important characteristic is that Fab A9E can distort both mZIKV and ImmZIKV particle structures.
Additionally, Fab A9E or IgG A9E LALA mutant can abolish or reduce the overall infection to myeloid cells when added to other infection enhancing antibody DV62.5:immZIKV complexes. Thus, antibody A9E represents a promising potential prophylactic and therapeutic candidate, as it is effective against all maturation states of Zika virus.
Abstract
Zika virus (ZIKV), a flavivirus, causes a range of clinical complications including microcephaly in human fetuses. Currently, there is no treatment or vaccine. Different maturation states (mature and immature forms) of flavivirus particles have been observed to be released from infected cells and are infectious. To understand how an ultrapotent human antibody (HMAb) A9E can neutralize these Zika particles, we determined the cryoEM structures of the A9E Fab fragment complexed with mature (mZIKV) and immature (immZIKV) ZIKV to 2.8Å and 7.5Å, respectively.
A9E binds to an epitope spanning Domain I (EDI), EDIII, and their linker in an E protein protomer in both immZIKV and mZIKV particles. A9E generally inhibited prior to or during virus attachment to cells, via virus aggregation, distortion of virus particles and inhibition of receptor binding.
ImmZIKV is particularly sensitive to structural distortion by Fab A9E. The primary mode of infection used by ImmZIKV is via antibody-dependent enhancement of infection (ADE)—the formation of virus complex with nonneutralizing or subneutralizing concentrations of antibodies, that leads to enhanced infection of Fcγ positive myeloid cells. IgG A9E, by itself displays poor ADE activity. When IgG LALA mutant or Fab A9E is added to other enhancing antibody (DV62.5):virus complexes, they can strongly reduce the overall ADE activity. This is likely due to their ability to distort virus particle structure, suggesting that HMAb A9E could be a potential prophylactic and therapeutic candidate against all maturation states of ZIKV."
Fig. 2 CryoEM Maps of the mZIKV complexed with Fab A9E (molar ratio of 3Fab:3E).