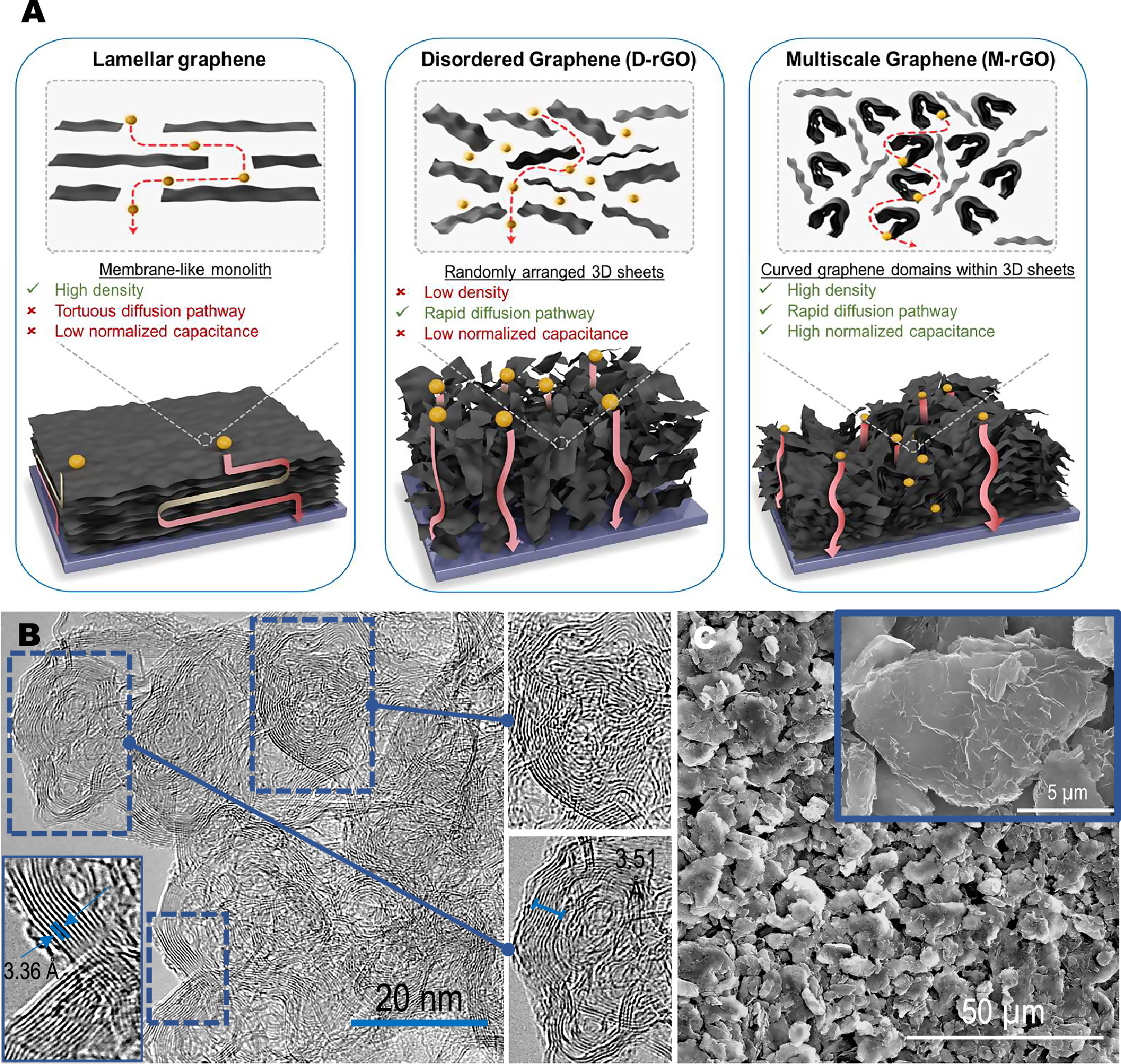
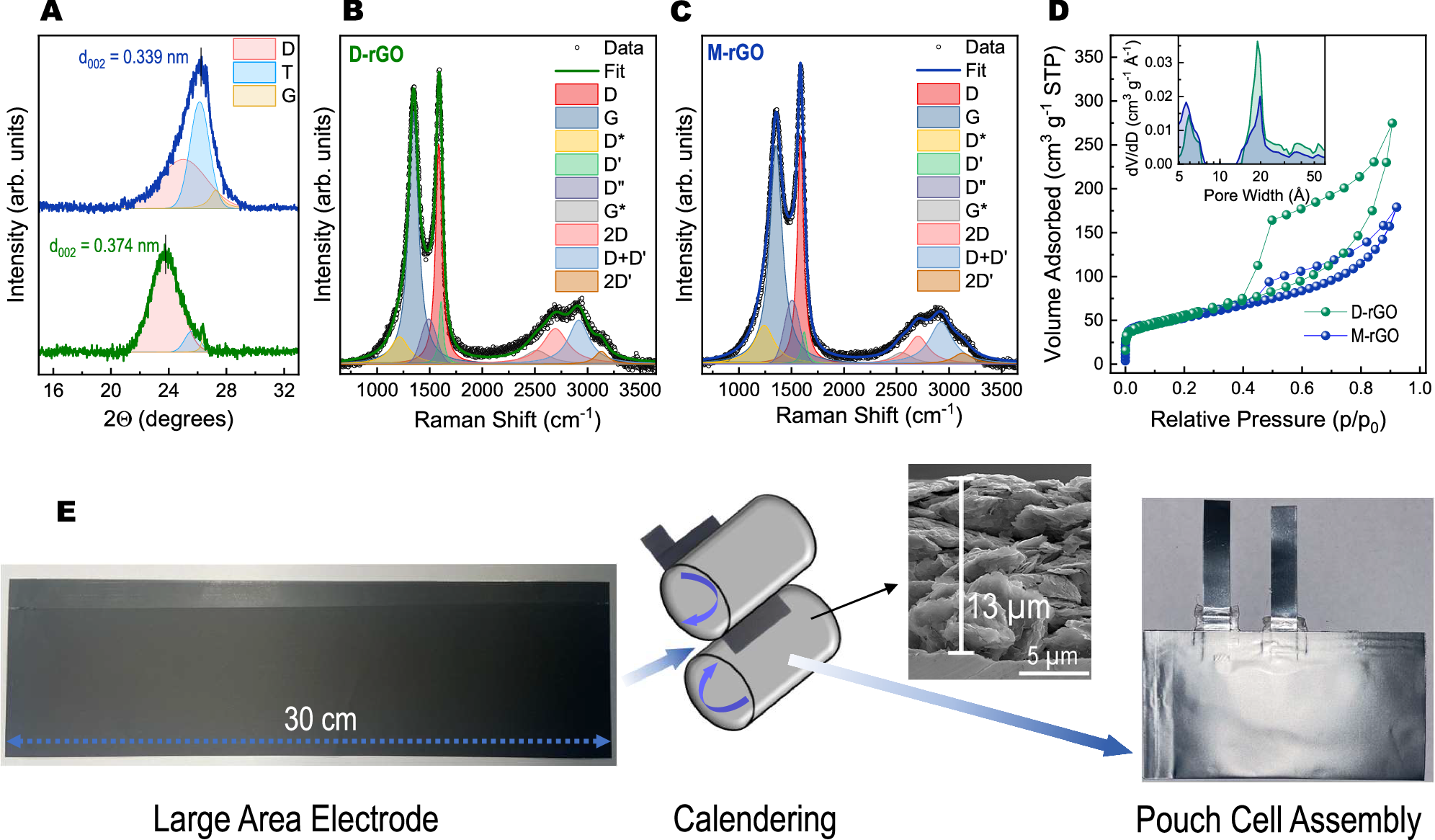
Long-term storage of electricity is still a huge challenge.
"... Promising LDES battery technologies
Vanadium flow
Vanadium flow batteries, developed ... in the 1980s, are now becoming popular around the world, with increased power and energy capacity
The world’s largest vanadium flow battery, a 175 MW/700 MWh system in Dalian, China, was developed by Rongke Power and completed in December 2024. Meanwhile, in the UK, a 5 MW array has been built which connects into the national grid system.
A vanadium flow battery stores energy in liquid electrolytes containing vanadium ions at four different oxidation states. The positive and negative electrolytes which are stored in separate tanks are circulated through battery stacks where the power conversion takes place. When charging or discharging, electrons transfer between the electrolytes through an external circuit, enabling energy storage and release without significant degradation.
Vanadium flow batteries can scale up easily, allowing a large the energy capacity for power supply for extended periods.
However, they have lower energy density than some other LDES options.
Lithium-ion
In contrast, lithium-ion batteries offer high energy density and fast response times, making them already popular for vehicles, consumer electronics and medical devices.
However, they degrade more quickly over time and may only last 500-3000 charging cycles before suffering noticeable capacity loss – compared to a reported 200,000 cycles for a vanadium flow battery.
There are also additional safety concerns with lithium-ion batteries related to thermal runaway leading to fires, while they are reliant on a scarce raw material and recycling is costly and complex.
Even so, the Hornsdale Power Reserve in South Australia and the Victorian Big Battery in Geelong both utilise lithium-ion Tesla Megapacks. The latter can store enough energy to power over one million Victorian homes for up to half an hour.
Iron flow
Iron flow batteries, which store energy in a liquid electrolyte typically made of iron, salt, and water, are an affordable and environmentally friendly option for long-duration energy storage.
These promise around 10,000 cycles with minimal degradation over time.
However, they have lower energy density than lithium-ion or vanadium flow and require more space for the same energy storage capacity.
Organic flow
Another potential option are organic flow batteries, which are still very much in the research phase, with carbon-based molecules being tested for use instead of metals such as vanadium or lithium.
While they may provide a cheaper, non-toxic energy storage solution, there are still big question marks about their energy density at scale and durability.
Limitations and challenges of LDES batteries
Despite their benefits, LDES battery development faces some significant challenges.
One major challenge is the high initial costs, with many of the technologies requiring substantial upfront investment in infrastructure, making large-scale deployment difficult.
LDES technologies, like other storage systems, experience energy loss during the storage and retrieval process, due to factors like self-discharge, internal resistance, and inefficiencies in the charging and discharging cycles.
Material and supply chain constraints also present significant issues for some LDES batteries. Certain technologies depend on rare or expensive materials, which can lead to supply chain vulnerabilities, as well as environmental concerns.
While many LDES technologies show great promise, some are still in the experimental phase and require further research and development before they can be widely adopted. ..."
Long-duration batteries can power a cleaner, reliable future